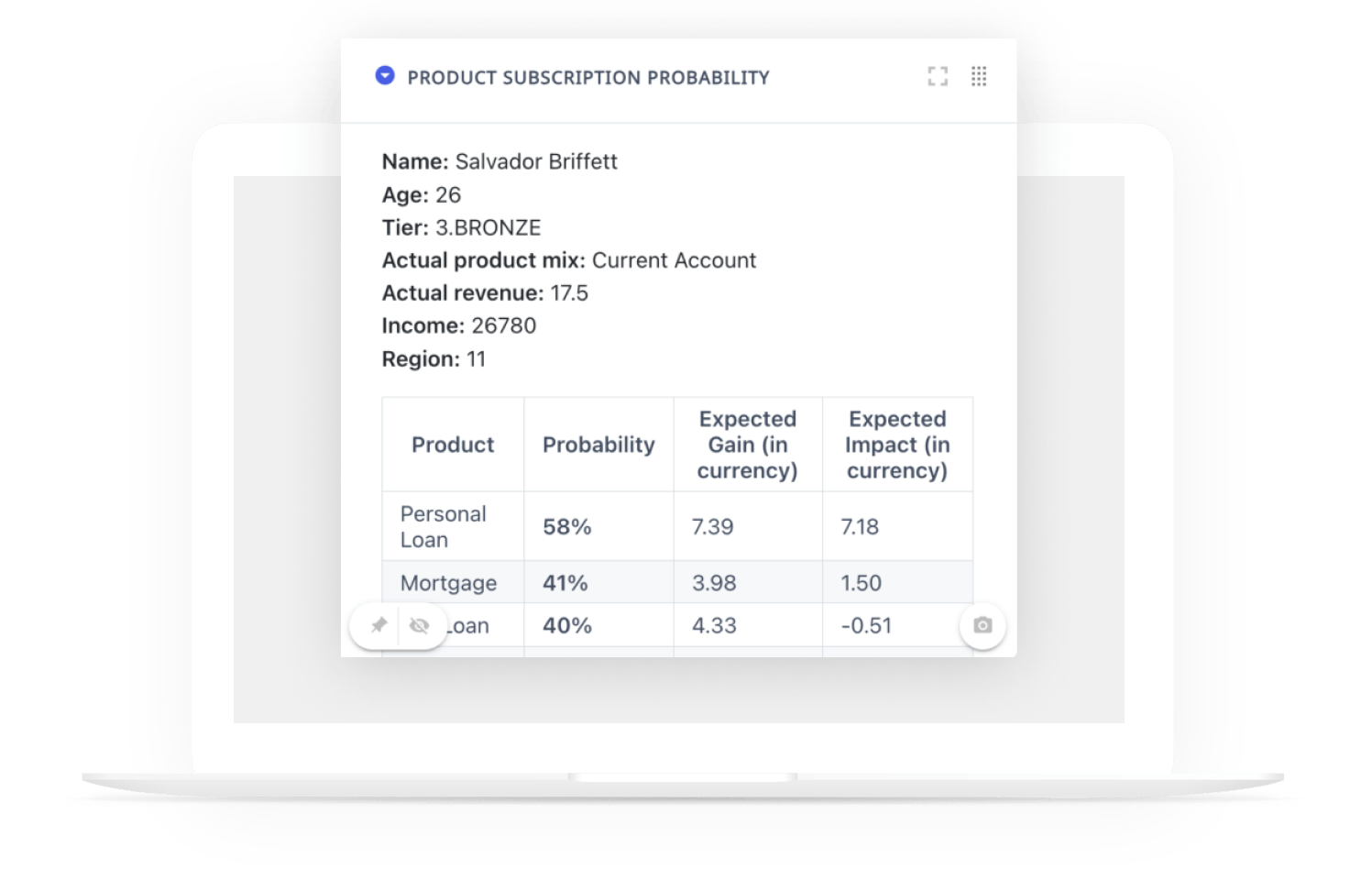
Clinical Site Intelligence
Easily uncover similar and/or competitive studies as well as clinical trial sites leveraged, run site reviews, identify selections in novel studies, and get access to sponsor overviews to guide data-driven decision making.
LEARN MORE