Easily map your Data Projects to the appropriate template
Easily access pre-built and customizable templates to centralize progress, sign-off, and other major KPIs on all GXP data and analysis projects.
Pharmaceutical, food, automotive and other goods manufacturing companies must follow Good “fill-in-the-blank” Practices (GxP) to ensure they bring safe and effective products to market. One of the many guidelines they have to comply with relates to the full traceability of data practices leading to a given decision – for instance, on running the appropriate clinical trials to validate a new drug, or the right tests to support development of a new car. Providing eased capacity to ensure traceability on roles, responsibility, quality, data project hierarchy and more becomes a critical accelerator for such companies.
Maintaining integrity of GxP data practices and proactively managing related risk is often manual and time consuming, with failure to comply with GxP guidelines resulting in fines, loss of market share and shutdowns. It is assessed that validation costs traditionally represent 25% of the total project cost.
Dataiku’s Streamlined GxP Workflows Solution, includes 6 customizable templates to provide visibility and oversight on roles and responsibilities of people working with data, the sensitivity and quality of that data, and the analysis actions taken at every step with centralisation of mandatory documentation and sign-off. By combining these governance templates with Dataiku’s unique platform capabilities, organizations with GxP processes can accelerate on both speeding time to value of all employees involved in data and analytics processes while reducing the burden of compliance enforcement.
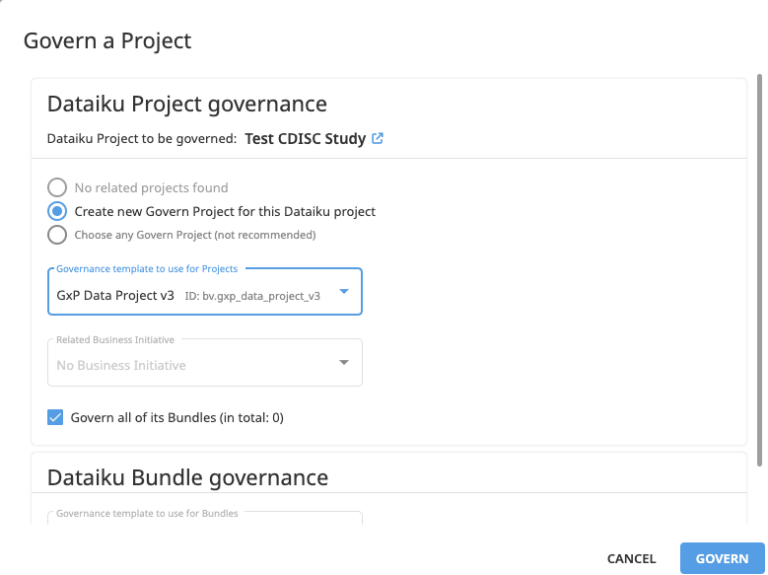
Easily access pre-built and customizable templates to centralize progress, sign-off, and other major KPIs on all GXP data and analysis projects.
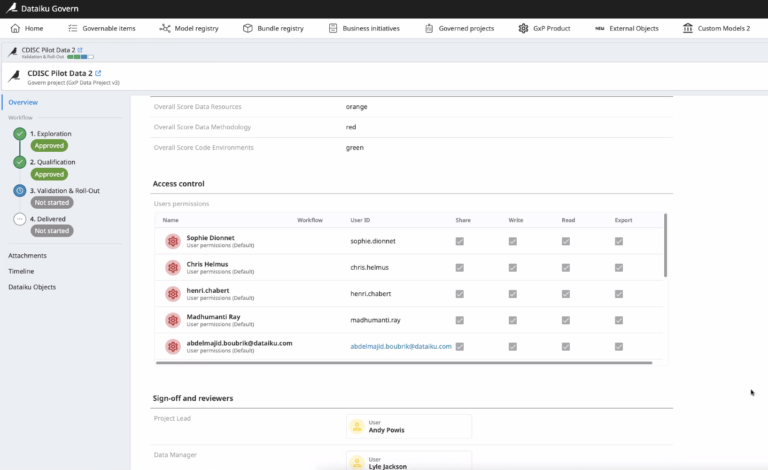
Review users’ access rights at a given time - eg. who would be able to write in the project.
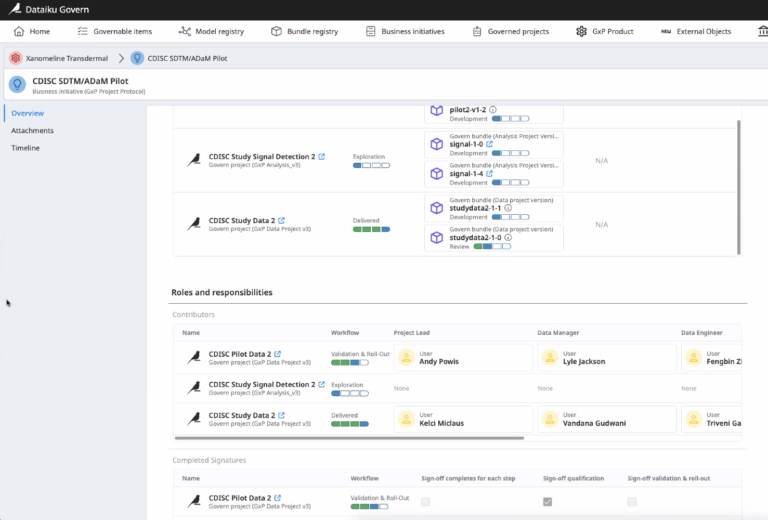
Understand users’ roles and permissions across Dataiku Projects and quickly identify enforced roles and responsibilities.
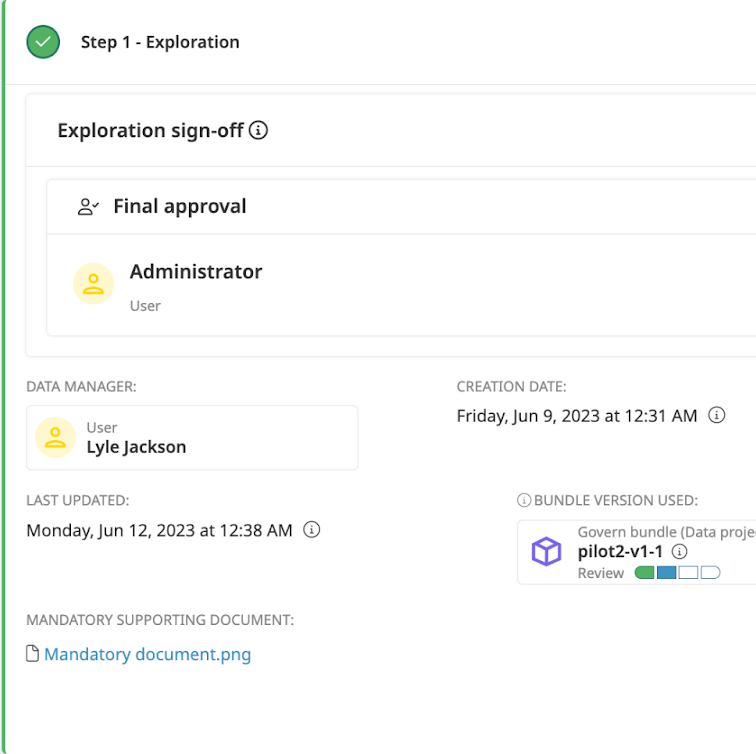
Ease centralization of key documents and versioning to ensure projects follow GxP guidelines throughout the development process.
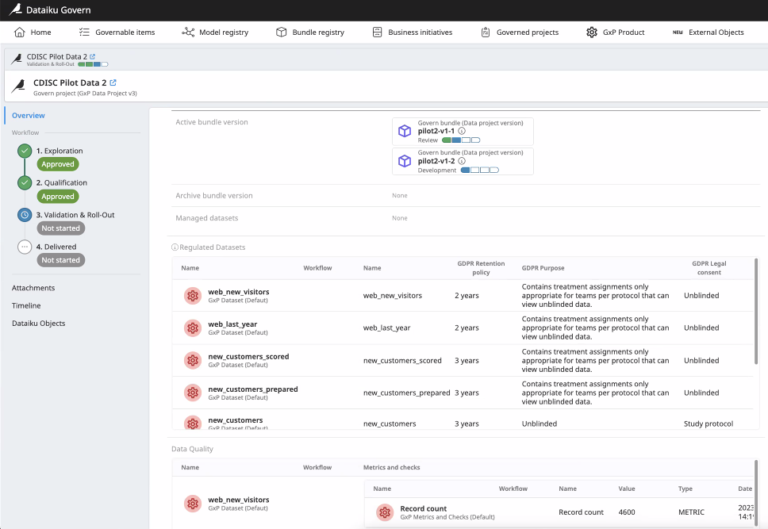
Simplify identification of the presence of sensitive data in the datasets used for a given data project, data quality metrics, as well as sign-off process.
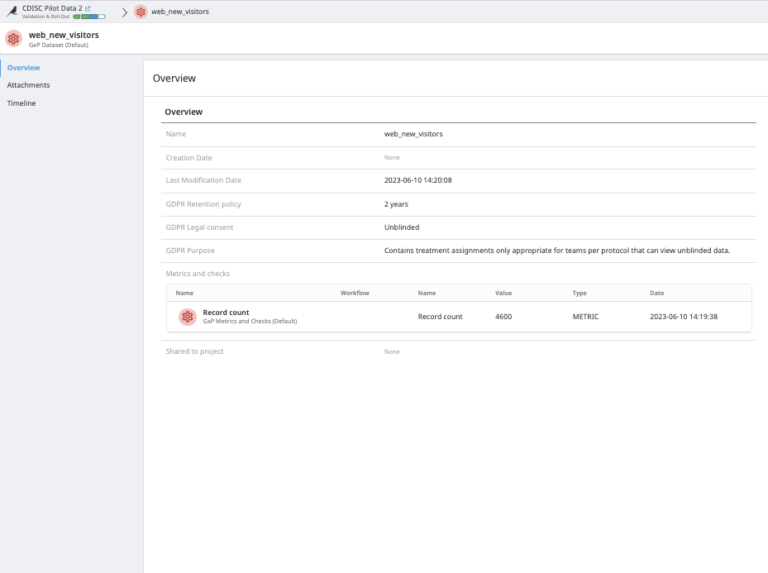
Ease visibility over the data quality metrics in the dataset used for a given data project.
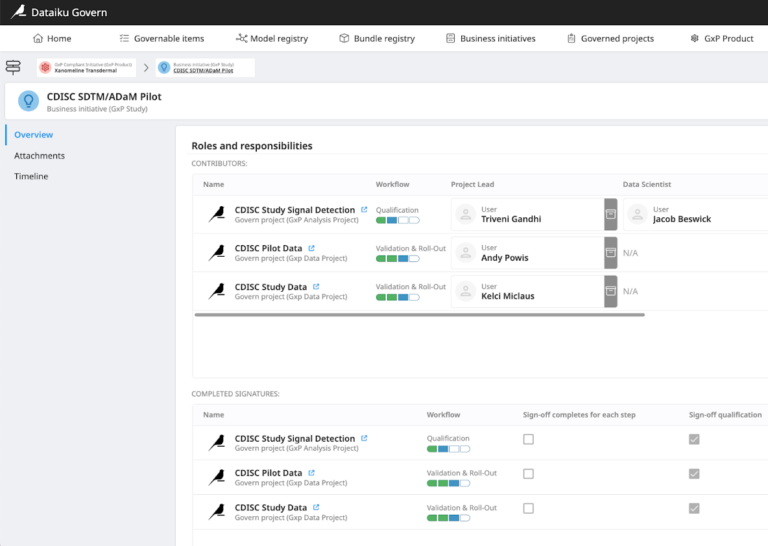
Facilitate oversight of the sign-off process for given data and analysis projects.