The End-to-End
Platform for Everyday AI
Build, deploy, and manage data and AI projects all in one place with Dataiku.
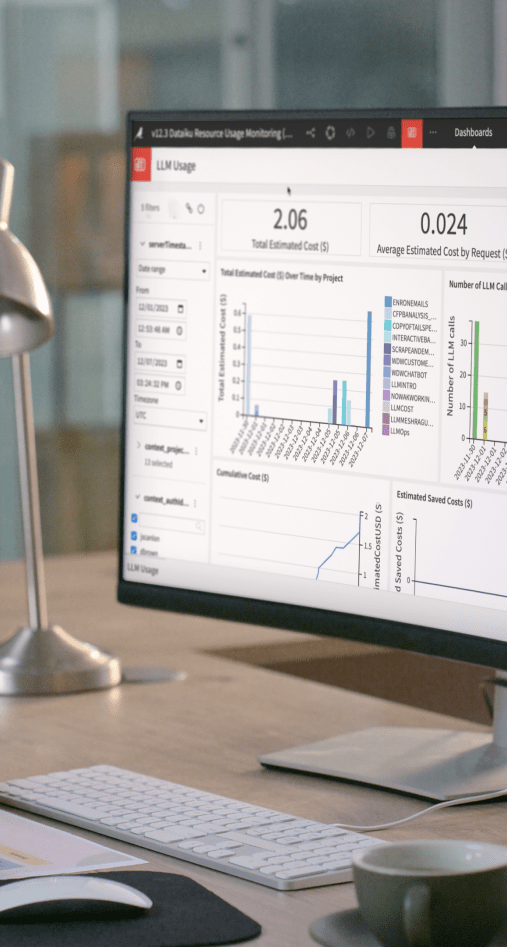
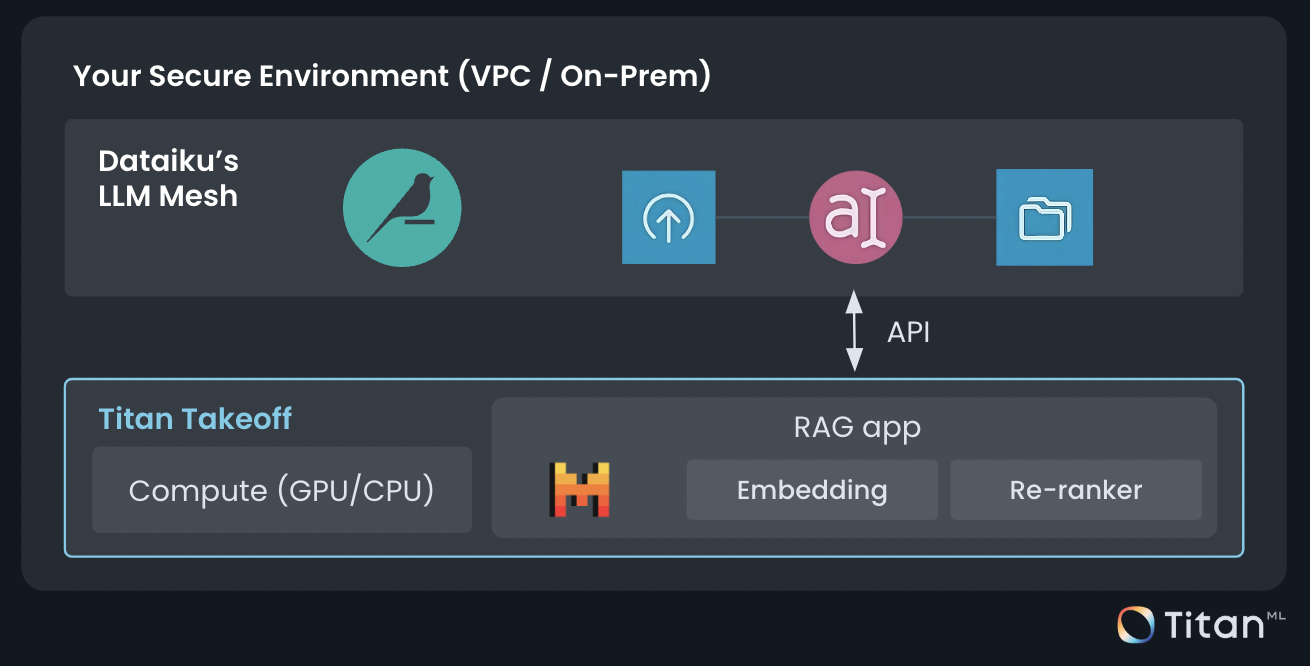
Generative AI
Move beyond the lab and build real and safe Generative AI applications at enterprise scale with development tools, pre-built use cases, and AI-powered assistants in Dataiku.
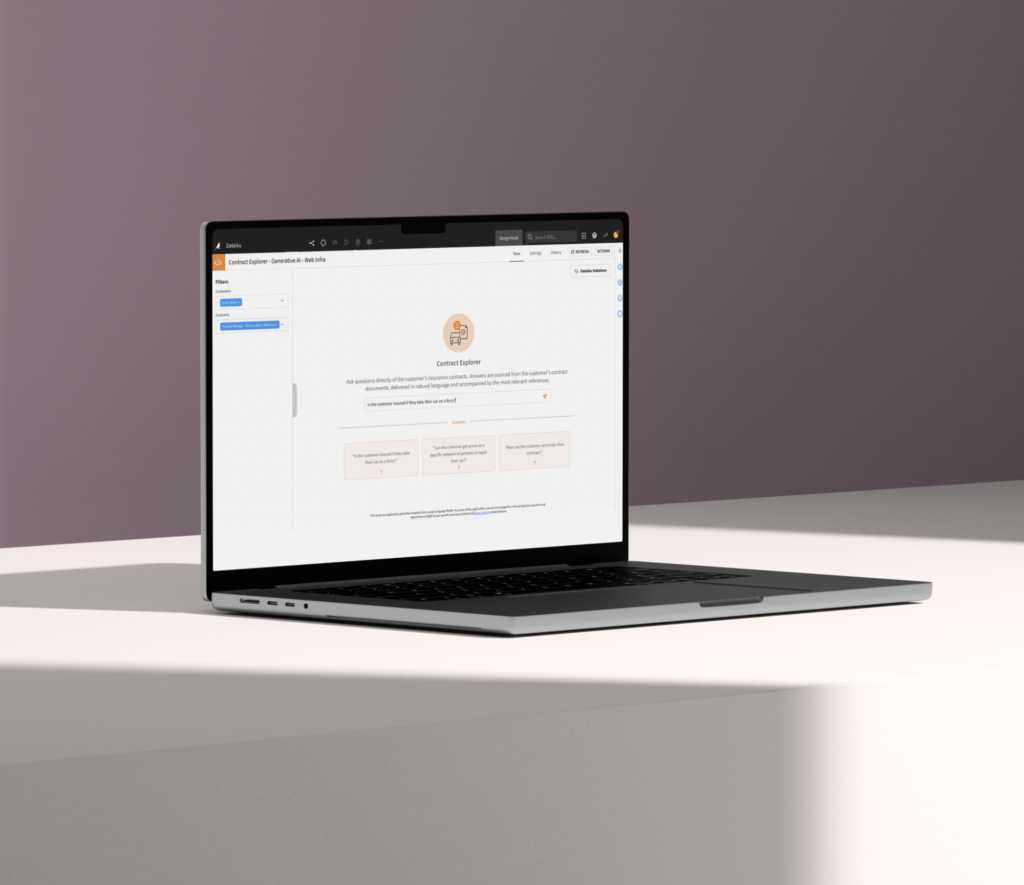
Data Preparation
Connect to, cleanse, and prepare data at scale. Dataiku provides both visual and coding interfaces in a shared space.
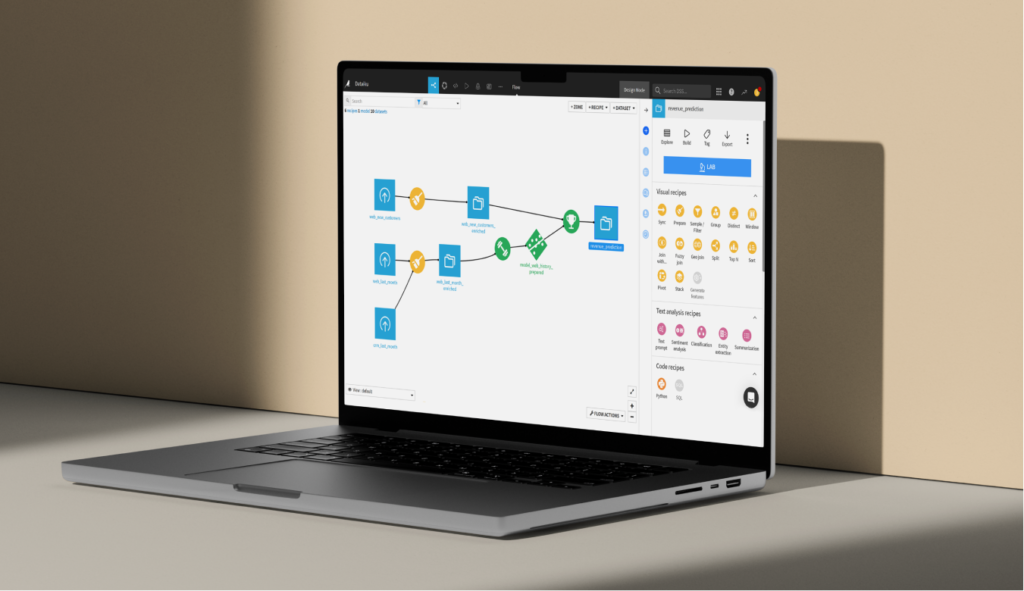
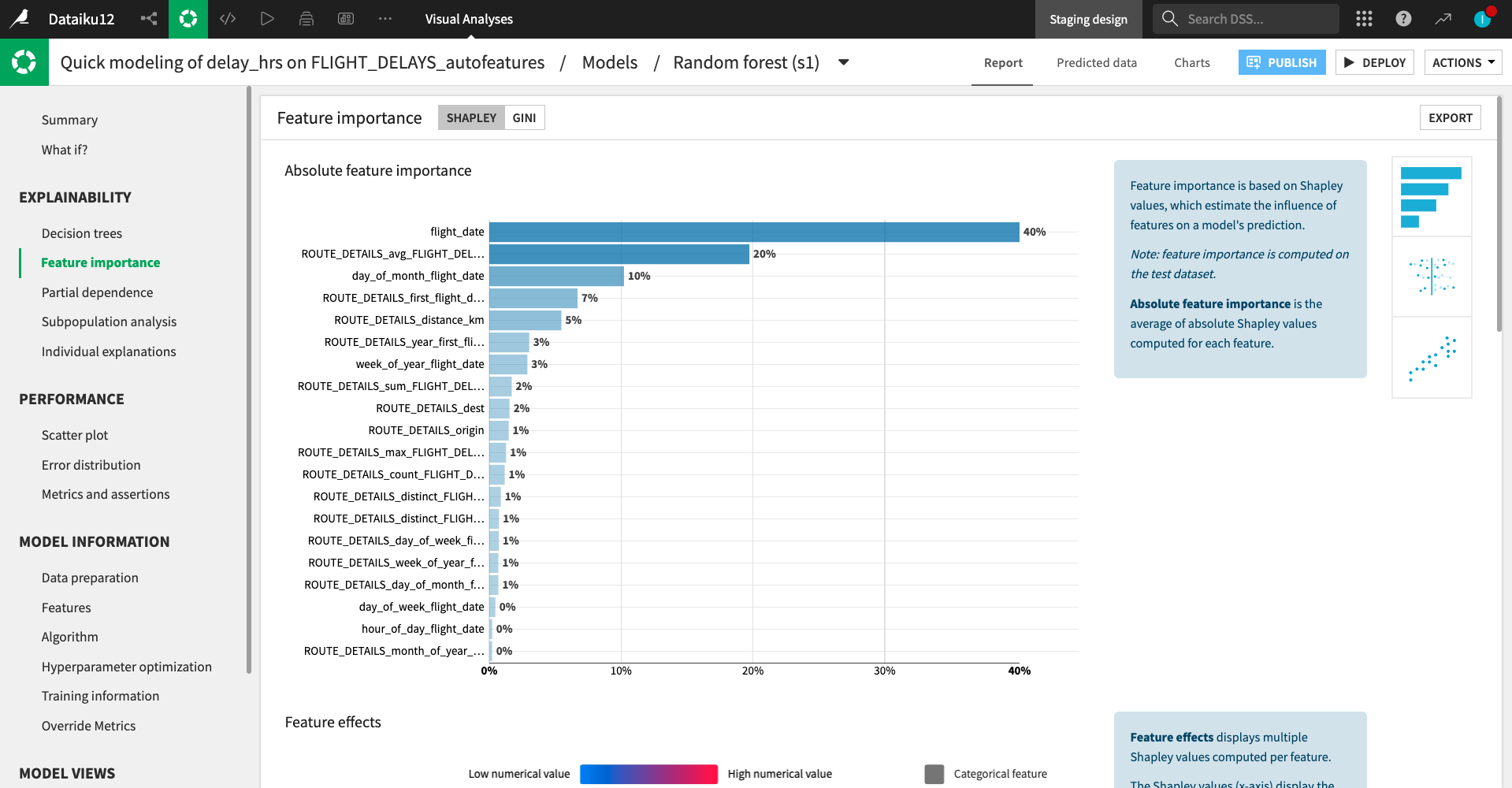
Machine Learning
Accelerate model building with Dataiku AutoML and a guided framework, or go custom with code.
Extensibility
Expand Dataiku’s native capabilities with plugins and custom applications for even more impact.

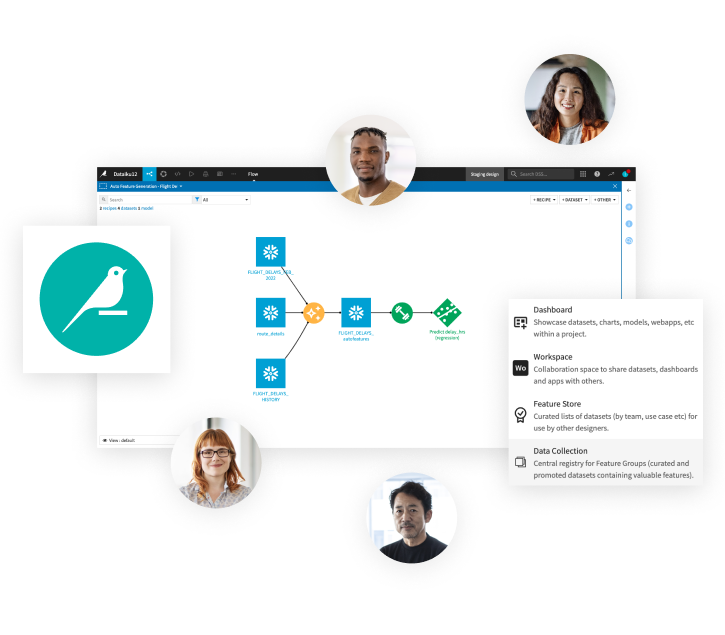
Collaboration
With Dataiku, teams can move beyond the lab and build real and safe Generative AI applications at enterprise scale.
Tailored Offerings for Every Team

Dataiku for AI & Machine Learning
Amplify the impact and productivity of data science teams with Dataiku’s end-to-end platform for AI and ML, bringing robust tooling for model design, development, and delivery.

Dataiku for
Data Analytics
Modernize enterprise analytics and accelerate time to insights with visual, cloud-based tooling for data preparation, visualization, workflow automation, and more.

Dataiku for
Enterprise AI
Drive innovation by unifying all people (from business to tech) and all enterprise data work (from analytics to Generative AI) in one centralized, governed platform for Everyday AI.
The Total Economic Impact™️
Of Dataiku
A composite organization in the commissioned study conducted by Forrester Consulting on behalf of Dataiku saw the following benefits:
reduction in time spent on data analysis, extraction, and preparation.
reduction in time spent on model lifecycle activities (training, deployment, and monitoring).
return on investment
net present value over three years.


Trusted by Global Enterprises

Assess Your Organization's
AI Maturity
Benchmark how your company compares to your peers and get tailored recommendations for increasing your AI maturity.